Research
- Home
- Research
Research Interest
Our Lab is interested in identifying the mechanisms by which innate immune cells, macrophages and microglia, drive multiple sclerosis (MS) progression. These cells, like other immune cells such as T- and B-cells, have specialized energy requirements which are not well defined in MS. Using cell culture-based assays and transgenic mouse models, we are invested in identifying key signaling pathways (pertinent to metabolic states) that allow these cells to become pathogenic/non-reparative during MS progression.
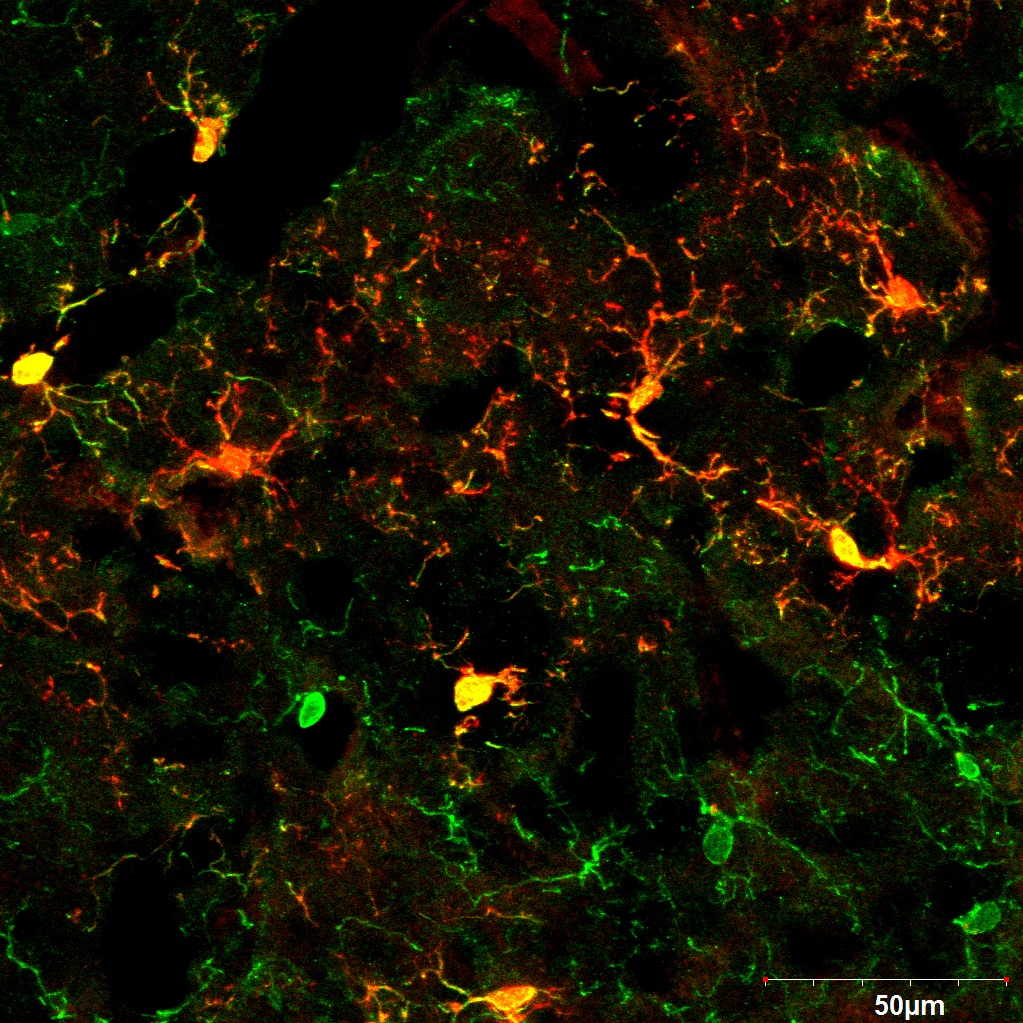
Microglia Metabolism in MS Progression
MS is an immune-mediated, demyelinating, and degenerative condition of the central nervous system (CNS) that targets the myelin sheath in the brain and spinal cord. This damage leads to physical disability and cognitive decline. In the early stages of MS, infiltration of peripheral immune cells plays a pivotal role. However, it is now well-established that the progression of the disease heavily depends on microglia, the brain’s resident macrophages. These cells are primarily responsible for clearing myelin debris, a critical function that diminishes over time, contributing to disease progression. This research focuses on understanding the metabolic requirements of microglia during MS progression. To study this, we utilize Experimental Autoimmune Encephalomyelitis (EAE), the most widely used mouse model of MS. By exploring the metabolic and functional dynamics of microglia, this work aims to shed light on potential therapeutic strategies to mitigate MS progression.
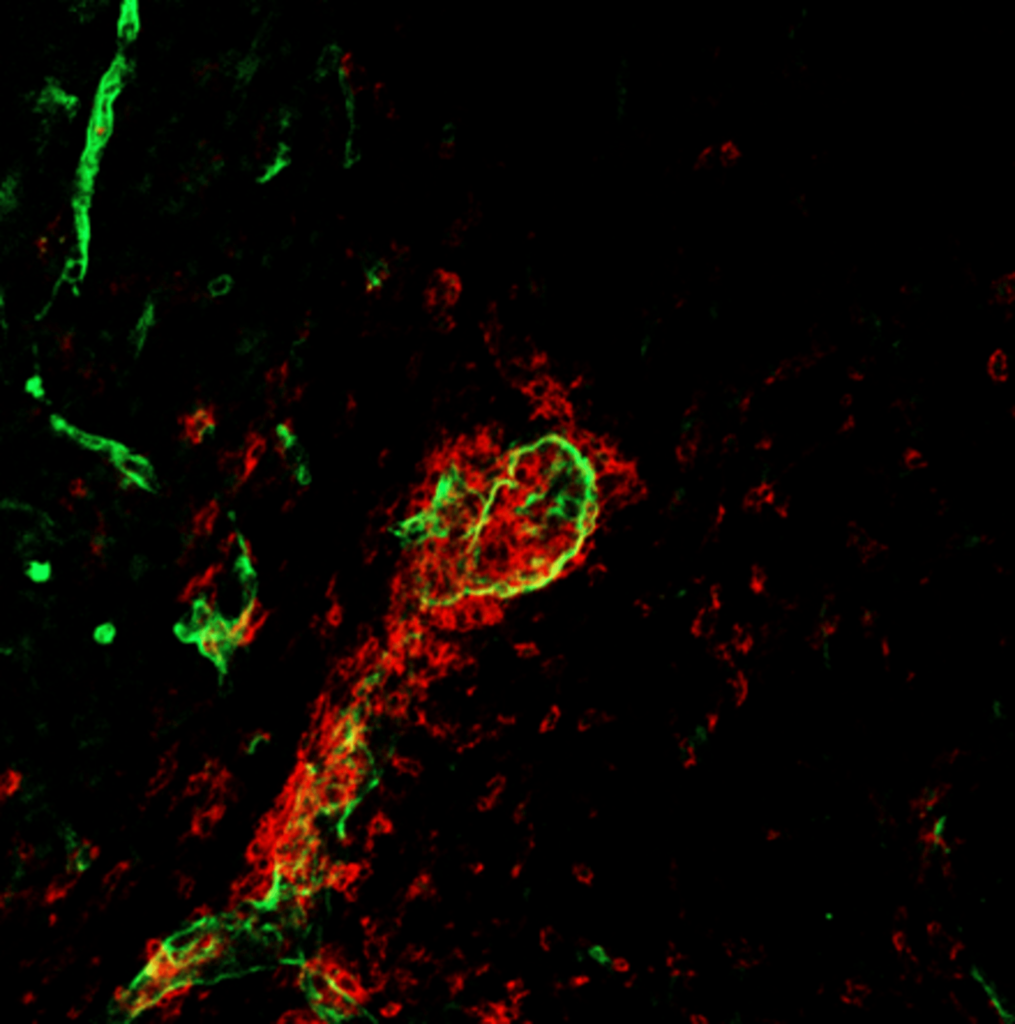
LPC (lysolecithin) Model of De- and Re- myelination
We established the LPC (lysolecithin) model of de- and remyelination in our lab to study demyelination and remyelination in the CNS. In this model, lysolecithin is injected into specific areas of the brain using stereotactic surgery to induce localized myelin damage, replicating certain aspects of demyelination observed in MS. Our research is particularly focused on exploring microglial immunometabolism to identify therapeutic targets that could slow disease progression, especially in aging mouse models. By understanding how microglial activation and metabolism contribute to neuroinflammation and neurodegeneration, we aim to uncover novel strategies to mitigate disease impact and enhance recovery.
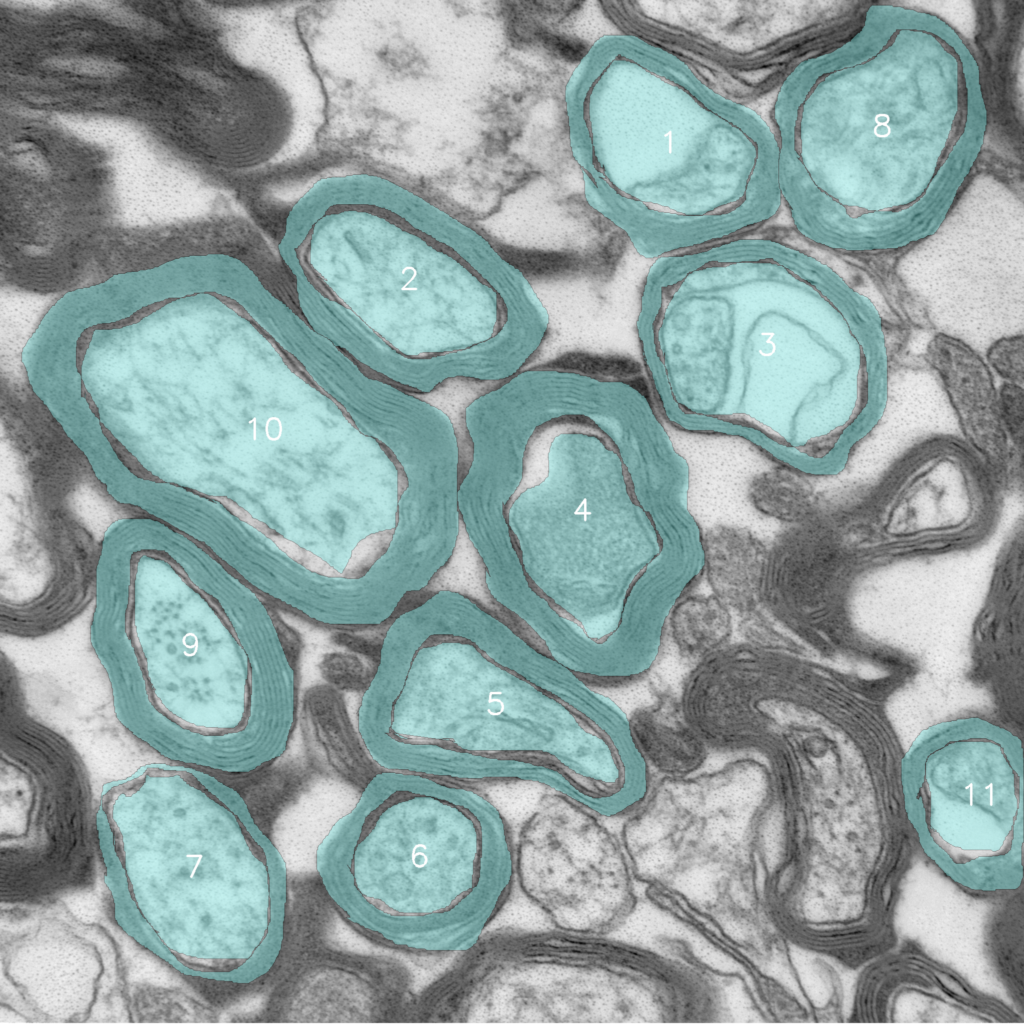
Microglial Metabolism in Brain Development
Microglia perform many critical homeostatic functions, including synaptic pruning. Synapses are structures that allow two neurons to connect and communicate effectively. During early brain development, neurons form more synapses than are required for proper brain functioning. Once the relevant synapses are established, microglia begin to ‘eat’ or ‘phagocytose’ the unwanted synapses in a process known as synaptic pruning. Aberrant synaptic pruning is associated with the formation of improper neuronal connections, which leads to social and cognitive challenges for mice and humans. Like other immune cells, microglia require specific metabolites supported by distinct metabolic pathways such as glycolysis or oxidative phosphorylation. However, the metabolic needs of microglia during the critical period of synaptic pruning are unknown. This research aims to investigate the metabolic pathways used by microglia during synaptic pruning and determine the impact of metabolic changes on microglia functions during the critical stages of brain development.
Thank you to our Funding Organizations






